Home | Glossary | Resources | Help | Contact Us | Course Map
Archival Notice
This is an archive page that is no longer being updated. It may contain outdated information and links may no longer function as originally intended.
Chemical Testing
Following the preliminary examination of an item thought to bear gunshot residues, a sequence of chromophoric (color-producing) tests should be performed. These tests may assist the visualization of residues that would otherwise be invisible and assist in determining a muzzle-to-target distance. These tests may also assist in shooting reconstruction.
| Note: |
|
The origins of a number of chemical tests are credited to the chemist Fritz Fiegl ( Spot Tests in Inorganic Analysis and Spot Tests in Organic Analysis) . These standardized tests originated as industrial spot tests for various elements and compounds; they do not need to be performed by a chemist. It is important to note that in the courtroom, issues may arise regarding
|
Up to three tests may be performed; where more than one test is performed, it is important that they are carried out in the following sequence:
- Modified Griess Test for nitrite compounds, a product of burning a nitrate, such as smokeless powder, or Reverse Modified Griess Test
- Dithiooxamide Test (rubeanic acid test) for cuprous (copper-bearing) materials
- Sodium Rhodizonate Test for all forms of lead residues or Bashinski Transfer Technique
The Modified Griess Test does not chemically interfere with the Dithiooxamide Test or the Sodium Rhodizonate Test. However, the Modified Griess Test must be performed first because sodium rhodizonate has the potential to interfere with nitrite testing.
Modified Griess Test
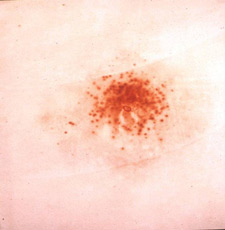
The Modified Griess Test is a chemically specific chromophoric (color-producing) test for the presence of nitrite compounds, such as those produced by the burning of smokeless powder. This test focuses on the analysis of the size and density of patterns of nitrite residues on a victims clothing or other objects as well as their reproduction at known distances using the suspects firearm in combination with the same type of ammunition.
This test has superseded the original Griess Test in that it does not involve the use of Marshalls Reagent, a solution suspected of being a carcinogen. Essentially, the new test avoids the use of N-(1-naphthyl)-ethylenediamine dihydrochloride, a component of Marshalls Reagent. This modification is safer, provides more vivid color results, and uses less expensive reagents and test media than the original Griess Test.
It is important to keep in mind that the Modified Griess Test does not yield a positive result for nitrate compounds, such as completely unburned smokeless powder particles. When smokeless powder burns or partially burns, nitrite compounds are formed, which may be detected by the use of the Modified Griess Test.
Chemistry
In the Modified Griess Test, a series of chemical reactions result in the conversion of any nitrite compounds that may be present on an item, such as victim clothing, into a bright orange dye in a chromophoric reaction. These dye pigments are preserved in an emulsion-coated test medium (desensitized photographic paper or printer paper) for later side-by-side comparison with known distance firings of similar residue patterns.
The chemistry of the Modified Griess Test is comprised of the following process:
- Nitrite residues are exposed to an acetic acid solution and heat to form nitrous acid.
- The nitrous acid combines with sulfanilic acid in the test medium to form a diazonium compound of sulfanilic acid.
- The diazonium compound couples with the alpha-naphthol (also in the test medium) to form a bright orange water-soluble azo (nitrogen-bearing) dye.
Additional Online Courses
- What Every First Responding Officer Should Know About DNA Evidence
- Collecting DNA Evidence at Property Crime Scenes
- DNA – A Prosecutor’s Practice Notebook
- Crime Scene and DNA Basics
- Laboratory Safety Programs
- DNA Amplification
- Population Genetics and Statistics
- Non-STR DNA Markers: SNPs, Y-STRs, LCN and mtDNA
- Firearms Examiner Training
- Forensic DNA Education for Law Enforcement Decisionmakers
- What Every Investigator and Evidence Technician Should Know About DNA Evidence
- Principles of Forensic DNA for Officers of the Court
- Law 101: Legal Guide for the Forensic Expert
- Laboratory Orientation and Testing of Body Fluids and Tissues
- DNA Extraction and Quantitation
- STR Data Analysis and Interpretation
- Communication Skills, Report Writing, and Courtroom Testimony
- Español for Law Enforcement
- Amplified DNA Product Separation for Forensic Analysts