Archival Notice
This is an archive page that is no longer being updated. It may contain outdated information and links may no longer function as originally intended.
Home | Glossary | Resources | Help | Contact Us | Course Map
Step Three: Removal of Denatured Proteins
Denatured proteins are removed during the extraction using a phenol chloroform isoamyl alcohol (PCI) combination. Phenol denatures proteins that are subsequently hydrolyzed with Proteinase K. Due to their negative charge, DNA molecules can be separated from other cellular components. Addition of PCI to a sample promotes separation of non-polar (organic) and polar (aqueous) phases. The phenol is not miscible with water, denatures protein, and sequesters the denatured hydrolyzed protein in the organic phase. During this process, the DNA remains in the aqueous phase in its double-stranded state.
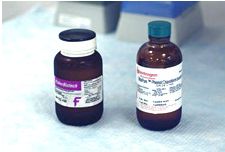
PCI may be purchased commercially or prepared in a ratio of 25:24:1. Because the pH of phenol (approximately 7.0) would generally be too acidic for purposes of DNA extraction, the phenol is buffered by saturation with TE buffer.
| Care must be taken to isolate only the aqueous phase during this procedure. Any residual PCI can compromise the filters in the filtration devices and also acts as a PCR inhibitor. |
Step Three Reagents
- Phenol
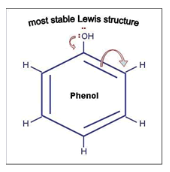
Phenol is a member of a class of organic compounds containing a hydroxyl group fixed to an unsaturated carbon in a benzene ring. Phenols are not true alcohols; they are more acidic than alcohols but less so than carboxylic acids. In most reactions they behave as nucleophiles. Phenols are also readily oxidized, more so than alcohols. This property is employed in DNA extraction. The addition of hydroxyquinoline to the reagent gives the organic phase an orange color, making it easier to differentiate the aqueous phase containing DNA. Polysaccharides and proteins are soluble in phenol, allowing for their separation from DNA.
| Although originally used for its antiseptic properties, phenol is highly toxic and should be handled in a fume hood while wearing personal protective equipment. Skin contact and inhalation should be avoided. |
- Chloroform
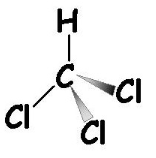
Chloroform (CHCl 3), or trichloro-methane, is a colorless liquid that is slightly water-soluble and miscible with organic solvents such as phenol. It is more dense than water or buffer (in which DNA is soluble), yet less dense than phenol. As it increases the phenol phase density, it promotes a sharp interface between the organic and aqueous layers. Chloroform also solubilizes lipids. During the extraction procedure, cellular debris that is not totally digested can be observed at the interface.
| Previously widely used as an anesthetic, inhalation of chloroform depresses the central nervous system. It is also a suspected teratogen and known carcinogen and should never be handled outside of a fume hood. |
- Isoamyl Alcohol
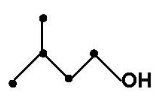
Isoamyl alcohol, or 3-methyl-1-butanol, is a primary alcohol. A liquid solvent, it is often included in genomic extraction protocols to help prevent foaming of the reagents, making it easier to detect the interface between the organic and aqueous phases. It is included in the protocol in very small concentration, compared to the other reagents.06
| A component of some paint strippers and other solvents, isoamyl alcohol causes irritation upon skin contact or inhalation. Vapors can also cause ocular discomfort and effects. This reagent should be handled with care and in a fume hood. |
Additional Online Courses
- What Every First Responding Officer Should Know About DNA Evidence
- Collecting DNA Evidence at Property Crime Scenes
- DNA – A Prosecutor’s Practice Notebook
- Crime Scene and DNA Basics
- Laboratory Safety Programs
- DNA Amplification
- Population Genetics and Statistics
- Non-STR DNA Markers: SNPs, Y-STRs, LCN and mtDNA
- Firearms Examiner Training
- Forensic DNA Education for Law Enforcement Decisionmakers
- What Every Investigator and Evidence Technician Should Know About DNA Evidence
- Principles of Forensic DNA for Officers of the Court
- Law 101: Legal Guide for the Forensic Expert
- Laboratory Orientation and Testing of Body Fluids and Tissues
- DNA Extraction and Quantitation
- STR Data Analysis and Interpretation
- Communication Skills, Report Writing, and Courtroom Testimony
- Español for Law Enforcement
- Amplified DNA Product Separation for Forensic Analysts